What is Hydrazine Hydrate?
Hydrazine Hydrate is one of the fuming, oily liquids, having no color. In terms of odor, it has similarity to ammonia. One can dissolve it in alcohol and water. However, in ethyl ether and chloroform, this HH is insoluble.
There are lots of applications of HH. Originally, it was used for rocket fuel at time of Second World War. However, in fungicides and herbicides manufacturing also, it is highly valuable ingredient. Other sectors, where we have found its use, include pharmaceuticals (tuberculosis drugs) and tranquilizers). At present, plastic industries also rely on Hydrazine Hydrate for creating the polymerization initiators and various blowing components.
Corrosion Reaction
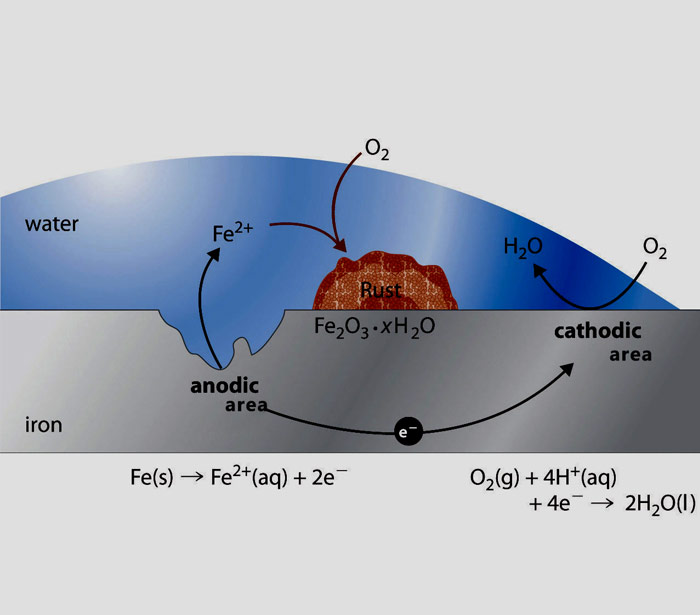
Corrosion is an electrochemical process, analogous to the operation of a battery. Any irregularity in a metal surface is a potential pit-forming anodic site. The metal acts as a conductor of electrons. The water is an electrolyte. But current will not flow if the cathodic site is polarized.
The reaction between the steel and the water liberates hydrogen, which forms a thin polarizing film on the cathode – unless oxygen is also present. When there is dissolved oxygen in the water, it combines with the hydrogen. The cathodic site is de-polarized, and current flows. When the corrosion current flows, ferrous ions go into solution. These ions may stay in solution as dissolved solids. Or, they ultimately combine with oxygen to form iron oxides, which can be deposited on the metal sur-face as rust. By eliminating dissolved oxygen, the oxygen scavengers permit the hydrogen film to be retained, polarizing the cathodic site and minimizing the corrosion current.
Accordingly, if you can remove the oxygen from water you can reduce the negative effects of corrosion. Our Product Remov -OX can be added to the boiler water that help to remove oxygen and so reduce the problems associated with corrosion.
Benefits of Hydrazine Hydrate
No Corrosive Solids
Remov -Ox completely miscible with water, reacts with oxygen to form nitrogen and water:
N2 H4 + O2 N2 + 2H2O
No solids are formed, and nitrogen is harmless to the system. Thus neither hydrazine itself nor its reaction products contribute dissolved solids to the system.
Compatibility with other treatment chemicals
Unlike other scavengers, Remov -Ox hydrazine’s’ reaction rates are not hindered by other chemicals such as phosphates, phosphonates, chelants or sludge conditioners, that are normally used in boiler-water treatment programs. This important property assures a more thorough protection of feedwater and boiler systems. It minimizes oxygen carryover to steam and condensate lines.
Reduced iron and copper pickup
Feed water often contains dissolved or finely dispersed iron and copper particles and their oxides. Hydrazine not only reduces rust to magnetite, but also reduces cupric oxide to its more passive form, cuprous oxide. Magnetite and cuprous oxide are dense particles, which are readily removed.In addition, considerable experience has shown hydrazine to be superior to sodium sulfite in controlling iron and copper pickup. Once hydrazine has promoted formation of a magnetite coating, boilers and other water systems remain largely free of metal oxide sludge. Hydrazine-treated boilers contain minimal metal oxide deposits when boilers are taken down for inspection and cleaning.
Control of pH
Water always includes small quantities of hydrogen and hydroxyl ions in equilibrium. With an excess of hydrogen ions present, an acidic condition exists. Iron is dissolved in the water; corrosion results. The rate of acidic attack is a function of pH. Since hydrazine is a base, it helps to neutralize the acid condition.
Under Pressure steam System
Our Hydrazine Hydrate [Remov – Ox] solution reduces oxidation in both low and high pressure steam systems. In fact, it is the best material that can be used for protection against oxidation in super critical boilers operating at pressures up to 3500 psi.They can be used throughout the steam-generating unit.
WHERE HYDRAZINE CAN BE USED ?
Hydrazine can be used to provide corrosion protection throughout the steam generating unit- in the preheater, boiler or condensate systems. It can also be utilized effectively in closed circulating hot water heating systems.
Feed water and preheat protection
To protect feed water and preheat systems, hydrazine can be fed to the storage sections of deaerators
Steam and condensate systems
Three factors are critical in assuring trouble-free operation: pH, amount of dissolved oxygen.
Boiler drums and tubes
Oxygen corrosion in boiler drums and tubes is prevented by normally maintaining hydrazine
Hot water heating
Hydrazine has been shown to be an effective corrosion inhibitor in closed hot water heating systems.
Industries Application
- Hydrazine hydrate monohydrateis a reductive agent used as materials of medicine, pesticides, dyestuff, blowing agent, and photographic developer.
- As an important raw material of Fine chemicals, hydrazine hydrate is mainly used as to synthesize the foaming agents.
- It also can be used as cleaning agents of deoxidize and decarbonation in boiler and agitated reactor.
- Hydrazine hydrate can be used to produce Antituberculotic and antidiabetic drugs.
- In farm chemical industry, hydrazine hydrate can be used to produce herbicide, Plant growth regulator, Insecticide agent, Sterilant, etc.
- Hydrazine hydratealso can be used as rocket fuel, nitrogen fuel, rubber auxiliary.
- Hydrazine hydrate has been used for the deproteination of the enamel. It may be used as a reducing agent in the following:
- Preparation of silver nanoparticles.
- Transformation of mono substituted nitrobenzene derivatives to the corresponding anilines.
- Along with graphite for the conversion of nitro compounds (aromatic and aliphatic) to the amino compounds.

